Introduction
- BOD stands for Biochemical Oxygen Demand, is the amount of oxygen required for bacteria to decompose biodegradable organic matter at certain temperature for a specific period of time.
- It is used mainly in determination of pollution strength of water in terms of requirement of oxygen, when this wastewater is discharged into water bodies.
- It is also useful in designing of wastewater plants and even this measures the efficiency of some of the treatment processes.
- The organic matter present in wastewater is grouped into two categories. They are –
a) Carbonaceous Matter –
This is the amount of oxygen required in sample for microorganisms to decompose the biodegradable carbonaceous matter. This is called 1st stage of decomposition.
b) Nitrogenous Matter –
This is called 2nd stage where nitrogenous matter get oxidised by autotrophic bacteria.
Note-
Dissolved Oxygen is the oxygen present in dissolved form in water as a Byproduct for aquatic plant photosynthesis.
Principle-
The sample is filled in an airtight bottle and incubated at specific temperature for 5 days. The dissolved oxygen content of sample is determined before and after five days of incubation at 20°C and then the BOD is calculated from the difference of initial and final DO. The initial DO is noted shortly after the dilution is made. After this measurement, whatever the amount of oxygen uptake take place is included in the BOD measurement.
Why is it needed ?
To know how much bacteria and organic matter is present in it and to evaluate what actually affects the water quality especially in sewage.
Apparatus Required –
- BOD incubator – It’s specificity is that it maintains the accurate temperature needed throughout the period of experiment.
- Burette and its stand.
- 300ml glass stoppers BOD bottles.
- DO Meter.
- Wash bottle.
- Conical flask.
- Measuring cylinder.
- Pipette with elongated tip.
- Gloves and seal starch.
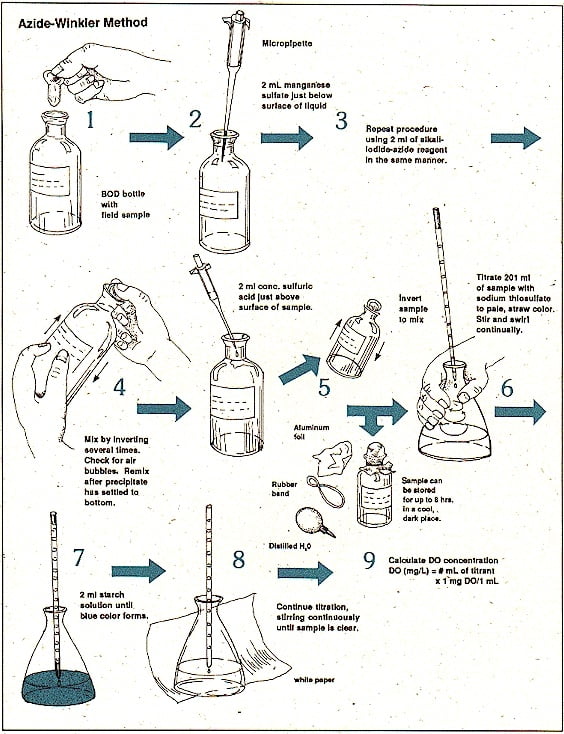
PROCEDURE –
- Take two bottles of sample from any river or pond or lake.
- Pour it into the brown bottles or BOD bottles.
- The blank solution is already prepared , so label the three bottles.
- Now, measuring the DO of each bottle and marking it as an initial reading.
- Closing the BOD bottles and seal the mouth of BOD bottles by seal starch, this is to avoid movement of air in and out of bottles.
- After sealing perfectly place all the samples in the incubator for 5 days at 20°C temperature.
- Making sure to wear gloves while performing the whole experiment.
- After 5 days, take out the samples and start to measure the final readings of DO in all samples.
- Also, taking care to clean the DO probe each time while taking readings of samples.
Stages of Decomposition of Organic Matter –
1st Stage –
- Here, the carbonaceous matter is oxidized.
- The B.O.D. at this stage is about 90% of total.
2nd Stage –
- The oxidation of nitrogenous matter takes place, that contain fats, proteins, amino acids, etc. by autotrophic bacteria.
Calculations –
The amount of oxygen required to decompose the organic matter in the given sample is calculated by the difference of the two DO levels.
BOD ( mg/l ) = ( D¹ – D² / V )
Where,
D¹ = initial DO reading.
D² = final DO reading.
V = Volume of sample.
Factors Affecting B.O.D. –
- Temperature
The increase in temperature results in increasing of plant growth rates, thereby leads in shorter lifespan of species and their overpopulation causing an algal bloom which reduces the levels of oxygen.
- Eutrophication –
This takes places because of over supply of nutrients like phosphorus and nitrogen. This results in explosive growth of plants and algae.
Why BOD test needs 5 days incubation period –
- This is because in 5 days duration 70 – 80% of organic matter gets oxidized.
- Another reason to take it as standard is to avoid the interference of nitrification of bacteria as this starts after 6th or 7th day.
- Therefore, by avoiding nitrification we get only “CARBONACEOUS BOD”.
- In India, people go for this test at the temperature of 27°C and within 3 days, so that results get earlier and also these are nearer to the actual values.
Limitation of BOD Test –
- It measures only biodegradable matter.
- Pretreatment is needed if any toxic waste is there in sample.
- It can’t be preferred for industrial waste.
- Long period of time is required to obtain the results.
Applications of BOD –
- Used in measuring waste loadings to treatment plants and also to check the BOD removal efficiency of such treatment systems.
- Used to determine the effects of discharges in receiving waters.
- It is very much useful in Water Analysis, in this BOD test is used for evaluating the aerobic destructibility of organic substances.
Conclusions –
- If the BOD will increase then it is because of present aerobic microorganisms.
- Here the oxidant used is microorganisms.
- The lower level of BOD indicates water is of good quality for aquatic life.
- The higher level of BOD indicates water is highly polluted and lower level of DO.
- If the sewage has high BOD then it will show anaerobic conditions.
References –
https://en.m.wikipedia.org/wiki/Biochemical_oxygen_demand#cite_ref-Sawyer_1-1
https://images.app.goo.gl/b1sEJVVRpjXTNbPx8
https://www.environmentechnology.net/mobile/jamesmacroony/biochemical-oxygen-demand
MLA. Metcalf & Eddy, Inc. Wastewater Engineering : Treatment and Reuse. Boston :McGraw-Hill, 2003.


