Introduction –
- Living things are made up of atoms, but in most cases, those atoms aren’t just floating around individually instead they are usually interacting with each other or (group of atoms).
- In our daily life, all the things we see, that holds them stiffly introduces us to “Chemical Bonds’’.
- All things are chemically bonded with each other. The Proteins we need, Carbohydrates we eat are all result of chemical bonding between atoms. Gas we use in our car is a result of Chemical bonding. Oxygen (O2) we breathe is a result of chemical bond. For that we must know what is a chemical bond?
- A Chemical Bond is defined as “the attractive force that holds two or more atoms in a molecule or an ion’’
- In this article, we’ll study Chemical Bond and its types in brief.
Why Chemicals bonds are formed?
It is because all the atoms trying to reach the most stable (lowest-energy) state. Many atoms become stable when their valence shell is filled with electrons or when they satisfy the octet rule (by having eight electrons). If atoms don’t have this arrangement, they will want to reach it by gaining, losing, or sharing electrons via bonds.
Types of Chemical Bonds –
- Ionic Bond.
- Covalent bond.
- Hydrogen bond.
- Van der Waals forces.
Ionic Bonds –
- It is Ionic bonds form between two or more atoms by the transfer of one or more electrons between atoms.
- It is also known as “electrovalent bond’’.
- Electron transfer that produces negative ions called “Anions’’ and positive ions called “Cations’’. These ions attract each other.
How Ionic bonding forms?
- The giving atom loses valence electron (2 or 3) so that bit has a full valence shell, but a positive charge.
- The gaining atom gives a valence electron (2 or 3) so that it has a full valance shell, but a negative charge.
- The negative and positively-charged ions are attracted to each other (like magnet) based on their opposite charge.
- If you look at the periodic table, you will see that ionic bonding usually occurs in compounds formed between one metal and one non-metal.
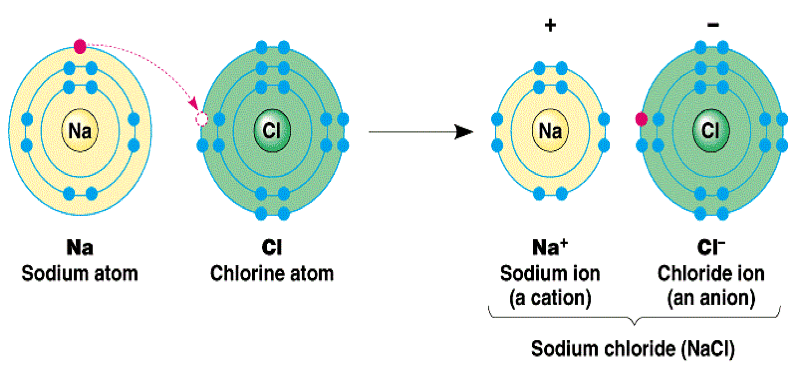
- Let’s examine the ionic bond in sodium chloride. A sodium atom, which has 11 protons and 11 electrons, has a single valence electron in its 3s subshell. A chlorine atom, has 17 protons and 17 electrons, has seven valence electrons in its third shell, represented as 3s23p5.
- In forming an ionic bond, the sodium atom, which is electropositive, loses its valence electrons to chorine. The resulting sodium ion has the same electron configuration as neon(ls22s22p6) and has a +1 charge, because there are 11 protons in the nucleus, but only 10 electrons about the nucleus of the ion.
Covalent Bond –
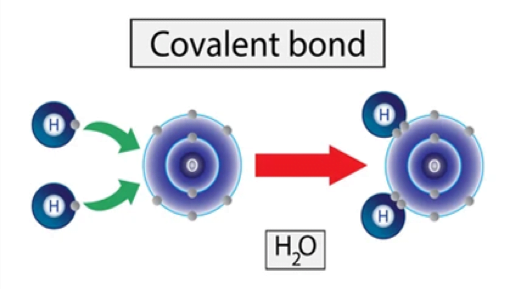
- It is type of chemical bond that links between two atoms in which electrons are shared between them.
- It is a chemical bond that involves the sharing of electron pairs between atoms. These electrons are known as shared pairs or bonding pairs and the stable balance of attractive forces between atoms when they share electrons is known as “covalent bonding’’.
- Let’s take an example of water molecule (H2O), there is a covalent bond between the oxygen and each hydrogen in water molecule (H2O). Each of the covalent bonds contain two electrons.one from a hydrogen atom and one from the oxygen atom. Both atoms share the electrons.
Types of Covalent Bond:
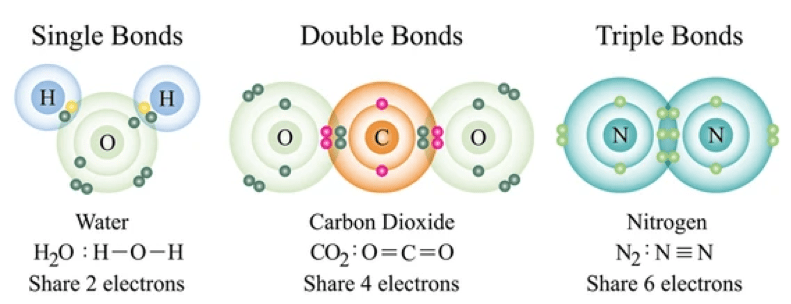
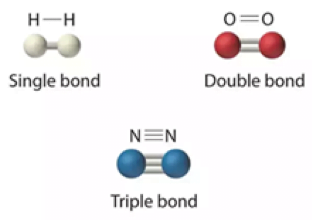
- Single Covalent Bond: –
- A bond formed by mutual sharing of one electron pair is called “single covalent bond”.
- It is denoted by single line.
- In chemistry, a single bond is a chemical bind between two atoms involving two valence electrons. That is , the atoms share one pair of electrons where the bond forms.
2. Double Covalent Bond: –
- A covalent bond formed by mutual sharing of two valence electrons is called “double covalent bond”.
- It is denoted by double short line.
3. Triple Covalent Bond: –
- A covalent bond formed by mutual sharing of three electrons is called “triple covalent bond”.
- It is denoted by triple short lines.
Types of covalent bond on the basis of polarity:
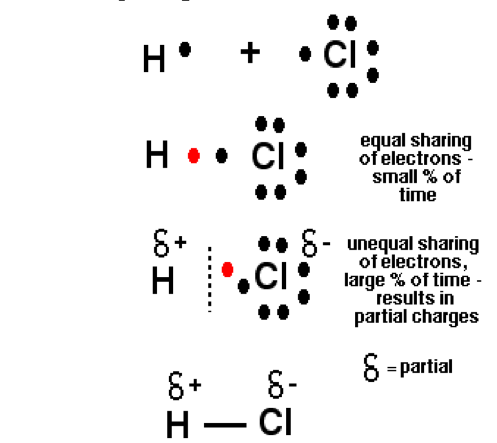
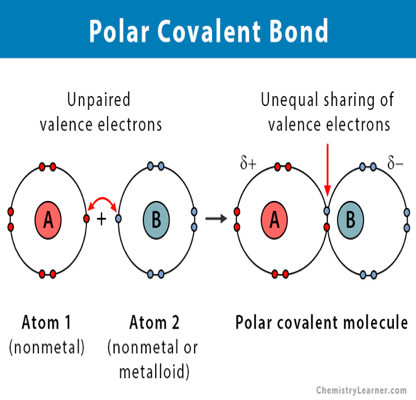
- Polar Covalent Bond: –
- A covalent bond formed between two different atoms is known as “polar covalent bonds”.
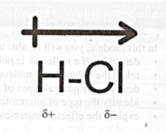
- For example, when covalent bond formed between H and Cl. Cl is more electronegative than H atom so electron cloud is shifted to Cl atom. This result partial (-) charge on Cl atom and partial positive charge on H atom respectively poles are formed therefore this type of covalent bonding is known as “polar covalent bonding”.
2. Non-Polar Covalent Bond: –
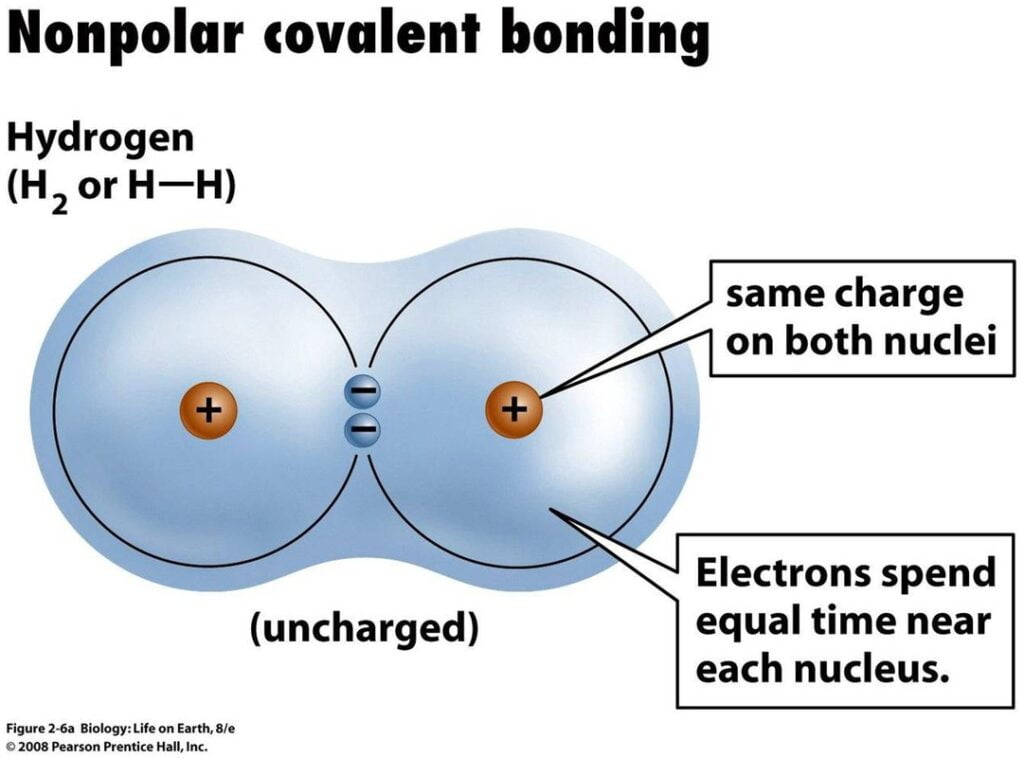
- A covalent bond formed between two alike atoms is known as “non-polar covalent bonds”.
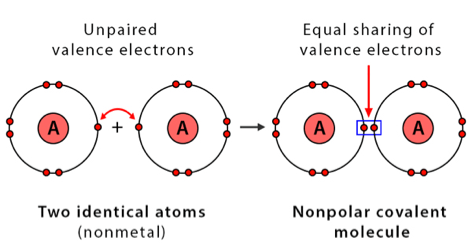
For example, as nuclei of each hydrogen (H) atom contains protons, the electrons in the bond are attracted to the nuclei. However, because the two atoms involved in a single covalent bond are both H atoms, each nucleus attracts the electrons by the same amount. Thus, the electron pair is equally shared by the two atoms.
Hydrogen Bond:
- Hydrogen has no inner shell electron and is very small in size, the positive charge density developed is high.
- The nucleus of hydrogen atom is exposed to attraction by nearby electron cloud, a lone pair electron on the electronegative atom.
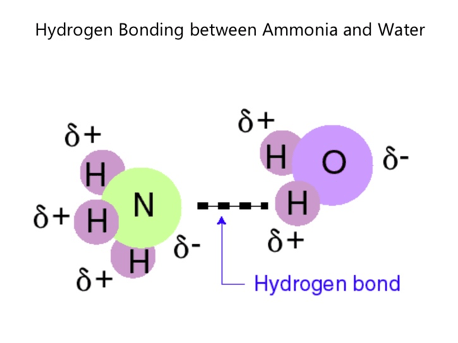
- Electrostatic attraction between hydrogen atoms bonded to small, strongly, electronegative atoms (N, O, and F) and the lone pair electrons on these electronegative atoms is called “hydrogen bonding”.
Hydrogen bonds are of two type: –
- Intermolecular Hydrogen Bond: – Hydrogen bond formed between two molecules. Intermolecular hydrogen bond is stronger than Van Der Waals forces.
- Intramolecular Hydrogen Bond: – Hydrogen bond formed between two different atoms in the same molecule.
Van Der Waals Forces –
- Van Der Waals Forces holds the molecules together which can be attractive or repulsive.
- It is named after “Johannes Diderik Van der Waals who invented the theory.
- They are weaker than normal covalent ionic bonds.
- They are additive and cannot be saturated.
- They have no directional characteristic.
- Van der Waals forces are independent of temperature except dipole-dipole interactions.
They are of following types: –
(a) Dipole-Dipole
(b) Ion-dipole
(c) Dispersion forces
(a) Dipole-Dipole: –
- Dipole-dipole forces are attractive forces existing between polar molecules (molecules that exhibit dipole moment), such as HCl.
- Because there is unequal sharing of electron between H and Cl atoms, partial positive and partial negative poles exist and are referred to as “dipole”.
- “The more electronegative atom becomes partially negative and has most electron density, whereas the least electronegative becomes partially positive and has a lesser electron density”.
- Because poles of opposite charges attracts, the partial negative and (i.e., Cl atom) will be attracted to the partial positive end (i.e., H atom) of another HCl molecule. This force is weaker than ionic and hydrogen bonds.
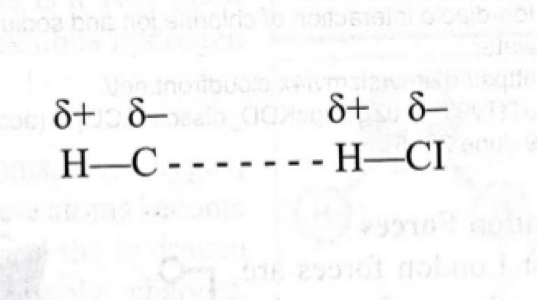
(b) Ion-Dipole –
- Exist in the attraction between a charged particle called ion (which can be a positively charged cation or negatively charged anion) and a polar.
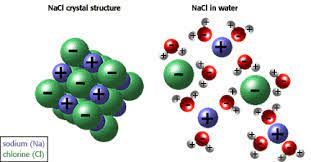
- When NaCl is dissolved in water (H2O), NaCl will dissociate into sodium cations (Na+) and chlorine anions (Cl).
- Because water is a polar molecule and has a partial negative and partial positive pole, the Na+ will form an ion-dipole interaction with the partial negative part of water molecule (O), while Cl– will form an ion-dipole interaction with the partial positive part of the water molecule (H+).
(c) Dispersion Forces or London Forces: –
- They are the weakest attractive force that are formed due to the temporary dipoles induced in non-polar molecules.
- This force is also called “induced-dipole-induced-dipole attraction”.
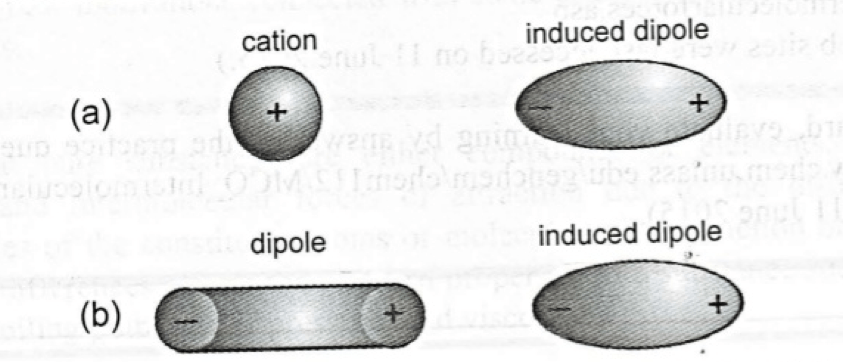
- For example, if there is an ion or polar molecule near a non-polar molecule, there will be a distortion of the nonpolar molecule due to the force exerted by the ion or the ion or the polar molecule. This distortion will result in temporary dipoles in the nonpolar molecules which is called “induced dipole”.
- Induced dipole is the separation of the positive and negative charges in a nonpolar molecule due to its nearness of an ion or polar molecule.
- On the other hand, when the induced dipole is due to the interaction between an ion and non-polar molecule, the interaction is called “ion-induced dipole interaction”.
- When the induced dipole is due to the interaction between a polar and nonpolar molecule, the interaction is called “dipole-induced dipole interaction”.
References –
https://www.nature.com/articles/s41598-018-36218-1
https://byjus.com/jee/covalent-bond/
https://www.sciencedirect.com/topics/chemistry/ionic-bond
https://www.slideshare.net/JeromeJerome1/intermolecular-forces-of-attraction