Introduction –
- Living matter is largely composed of biomolecules consisting of water and complex polymers of proteins, lipids, Nucleic acids and carbohydrates.
- Carbohydrates are compounds of tremendous biological importance. They store and produce energy, the ribose sugar is an important component of nucleotides, they are associated with proteins and lipids forming glycoproteins & glycolipids respectively; they are involved in forming structural features or organelles of cells. They are also found involved in oxidation of lipids.
- Considering their biological function, carbohydrates are crucial component of healthy and balanced diet. They provide us energy for daily tasks and are primary fuel source for our brain’s high energy demands.
- Chemically, carbohydrates are molecules that contain elements like carbon, hydrogen, and oxygen atoms in specific ratios.
What are Carbohydrates?
- They are broadly defined as polyhydroxy aldehydes or ketones or substances that hydrolyze to yields one of these compounds. The word Carbohydrates means “hydrates of carbon”.
- Carbohydrates are represented with formula C(H2O))Y
- They are probably the most abundant and widespread organic substances in nature.
- About 64% of the foods in our diet consists of carbohydrates.
- They are soluble in water and due to its sweet nature it is also referred to as “saccharides” (Sweet sand in Latin) .
- They are synthesized by plants by photosynthesis process, from carbon dioxide and water.
6CO2 + 6H2O → C6H12O6 + 6O2
Why do we need Carbohydrates?
- Carbohydrates are said to be body’s main source of energy: Their metabolism produces ATP which acts as a fuel to power brain, kidneys, heart muscles, and central nervous system. Each gram of carbohydrates gives 4-gram calories.
- Our body can store extra carbohydrate in our muscles and liver and utilized the stored carbohydrate when we are starving.
- A carbohydrate-deficient diet may cause headaches, fatigue, weakness, difficulty in concentrating, nausea, constipation, bad breathing and vitamin and mineral deficiencies.
- If a person does not have a good supply of carbohydrates, their body will utilize protein and fats as an energy source. However, as protein is crucial for so many other essential functions, such as building and repairing tissues, the body prefers not to use it for energy production.
Where do Carbohydrates come from?
- Most carbohydrate are from plant origin. They produce carbohydrate by photosynthesis using carbon dioxide, and water in presence of sunlight.
- All plants contain carbohydrate, which represent a significant portion of our dietary intake.
- We can get carbohydrate from animal source like meat, milk and Egg.
- The major carbohydrate such as sugars, maltose, fructose and sucrose obtained from plants whereas lactose is found on milk.
Functions of Carbohydrates –
- One of the primary functions of carbohydrates is to provide energy to the body.
- Fibrous carbohydrates promote Digestive Health.
- They provides carbon atoms for the synthesis of other biochemical substances such as (proteins, lipids, and nucleic acids).
- Carbohydrate helps in formation of part of the structural framework of DNA and RNA i.e. nucleotides.
- Carbohydrate that are linked to lipids are structural components of cell membranes.
- Carbohydrates that are linked to proteins function in a variety of cell-cell and cell-molecule recognition processes.
- Carbohydrate (as glycoproteins and glycolipids) have contribution in the structure of cell membrane and cellular functions such as cell growth, adhesion and fertilization.
- They are structural components of many organisms, which includes the fibre (cellulose) of plants, exoskeleton of some insects and the cell wall of microorganisms.
How does the body process ingested Carbohydrates?
- The body breaks down complex carbohydrates to simple one and made it available in the blood stream. The pancreas releases insulin to help move glucose into the body’s cells, which can be further used or stored
- The cells takes glucose from blood via glucose transporters.
- The glucose is metabolized by glycolysis, followed by Kreb’s cycle.
- The NADH produced during glycolysis and Krebs cycle, enter Electron Transport Chain (ETC).
- The NADH acts like electron donor to electron transport chain. Electron Transport Chain is a series of protein complexes that transfer electron from donor to acceptor by oxidation and reduction reactions. During the redox reactions, protons are transferred across the membrane and gets accumulate in inter-membrane space of mitochondria. It causes electrochemical gradient formation across the membrane which drives ATP synthesis by ATP synthase enzyme using oxidative phosphorylation.
- If the body is already storing enough energy and does not require more, it converts the glucose into fat, which can lead to obesity.
- When Glucose exceeds than optimum level it become toxic and can damage the body.
- Insulin is responsible for preventing a person’s blood sugar levels from getting too high in the body
- A diet which contains lots of sugary foods and carbohydrates can cause too much reliance on the insulin response, which may lead to health issues such as diabetes or obesity.
Classification of Carbohydrates–
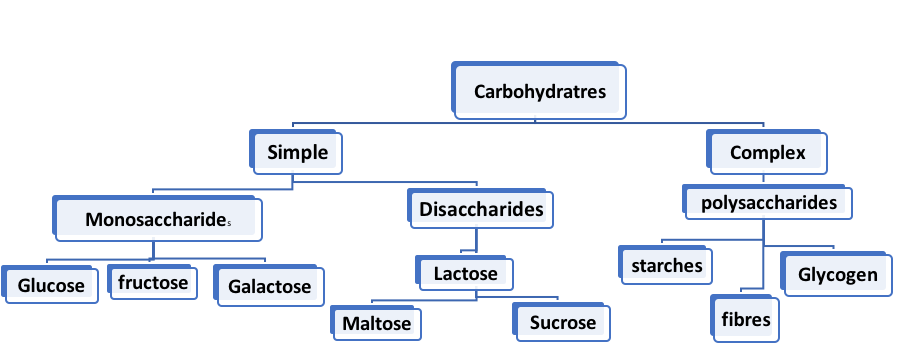
Carbohydrates are mainly of two types: –
- Simple Carbohydrates
- Complex Carbohydrates
- Simple Carbohydrates –
- Simple carbohydrates are mostly involved in producing energy instantly. It includes monosaccharides and disaccharides.
- Simple sugars can be natural or the processed one. Carbohydrates present in soft drinks, candy, cookies and other sweet snacks are form of processed sugar.
- Natural and simple carbohydrates are found in Fruits, milk and vegetables. Honey is a natural sugar as well.
2. Complex Carbohydrates: –
- Complex carbohydrates are involved in structural and functional role.
- They acts like store house for producing.
- Complex carbohydrates are polymers of monosaccharides, held together by chemical bonds.
The Simple and Complex carbohydrates are further classified as follows-
- Monosaccharides (simple carbohydrate).
- Disaccharides (simple carbohydrate).
- Polysaccharides (complex carbohydrate).
Monosaccharides: –
- Monosaccharides are simple sugars that contains only one oxygen atom and two hydrogen atoms for each carbon atom present in the molecule. They are monomers or repeating units of polysaccharides or complex carbohydrates.
- General formula of monosaccharides is (CH2O)n.
- Monosaccharides are “reducing sugars”. Because of its free functional aldehyde or ketone group which behaves like a reducing agent.
- The reducing ability of sugar can be tested by Benedict’s test.
- Monosaccharides are sugars, with sweet taste, and are soluble in water and insoluble in non-polar solvents.
- They are further classified on the basis of number of carbon atoms present in each molecule as trioses (3C), tetroses (4C), pentoses (5C), hexoses (6C), heptoses (7) and so on.
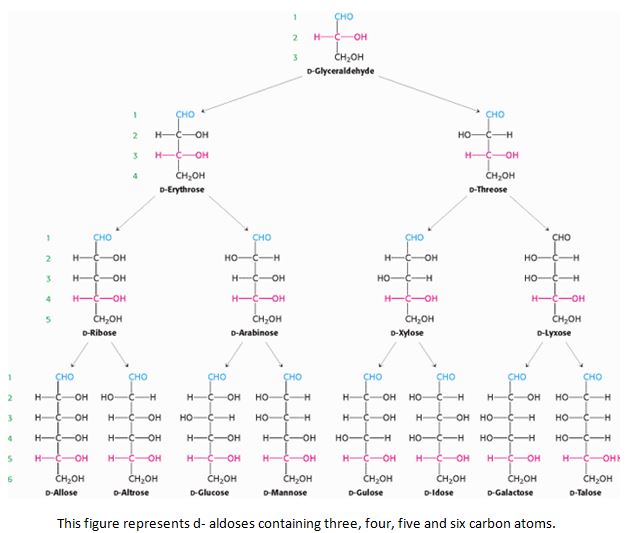
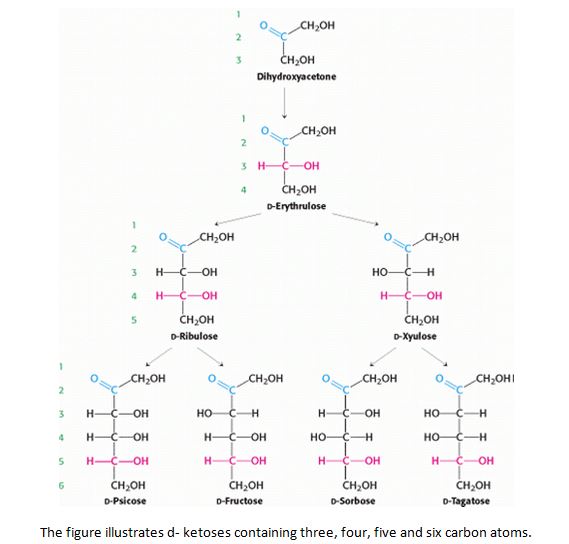
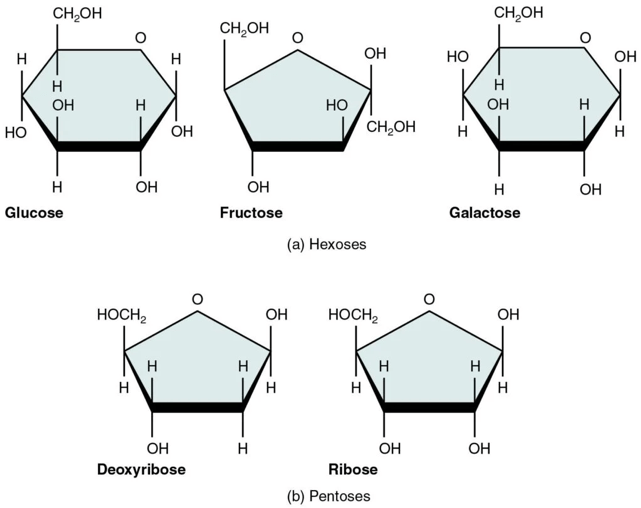
The nomenclature (names) all sugars ends with ‘-ose’ example glucose, galactose, mannose and sucrose.
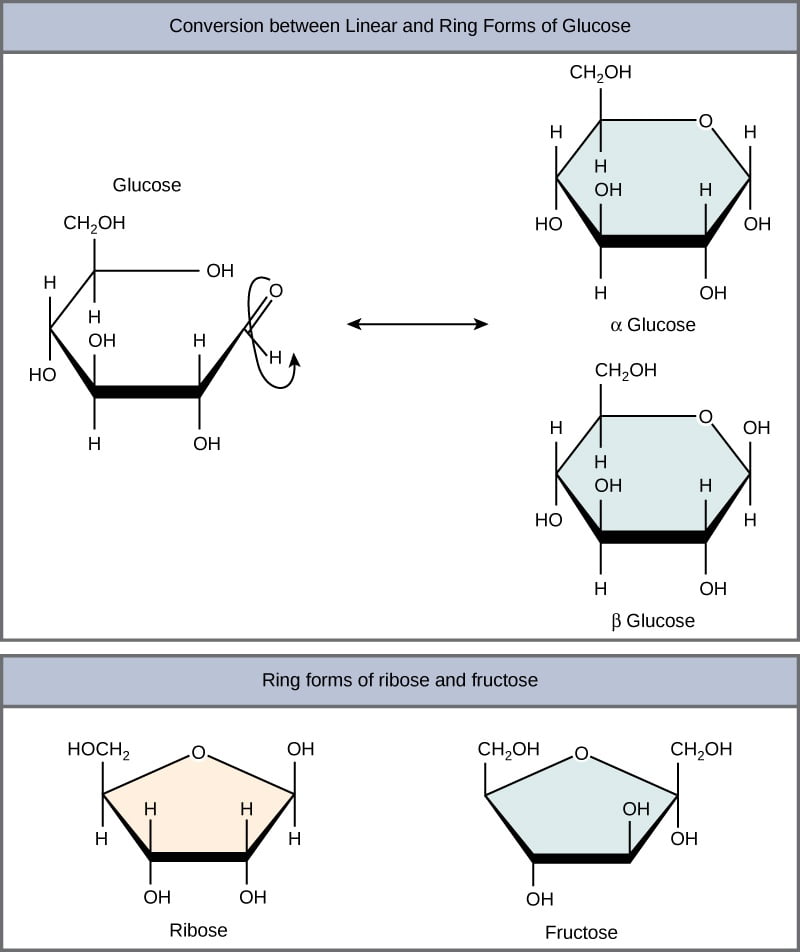
They are in either straight (linear) chains or in the ring/cyclic forms. The straight chain and ring form are interchangeable structure. When the straight chain form a cyclic structure, it can result in the formation of alpha and beta form of monosaccharides. The alpha and beta are named based on the orientation or position of hydroxyl group at C1 (anomeric carbon). If the hydroxyl group is above the plane of C1 it is called as alpha and if it is below, it is called as beta.
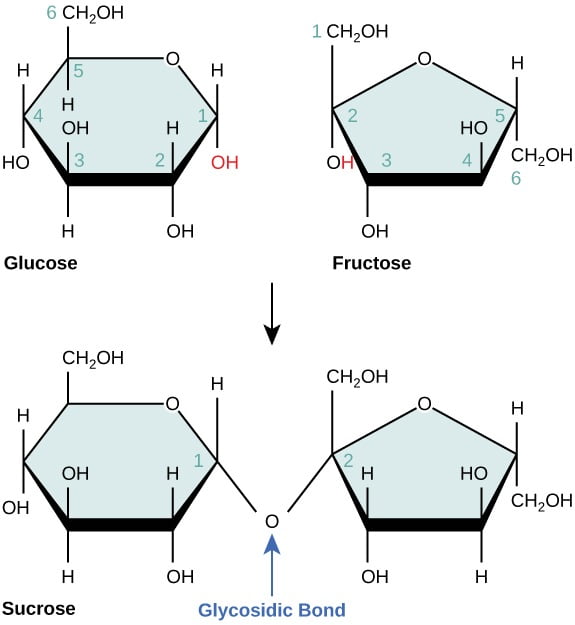
2. Disaccharides –
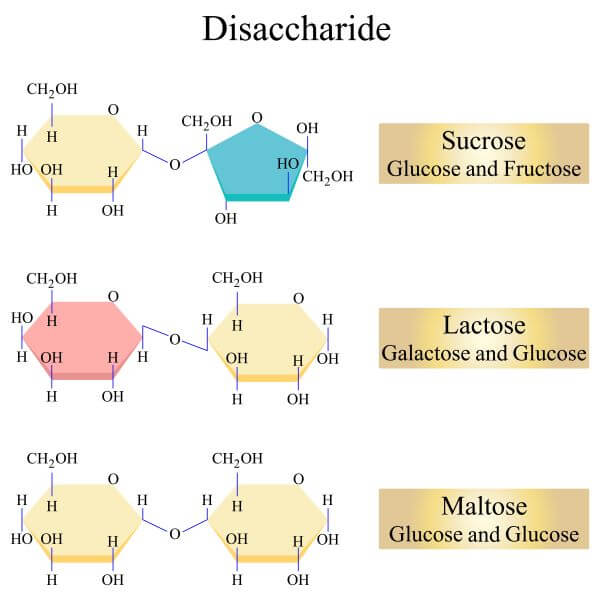
- Disaccharides are made up of two monosaccharides bonded together by a Glycosidic bond.
- The Glycosidic bond is formed by the condensation reaction and releasing a water molecule.
- Disaccharides can split apart into two monosaccharides by hydrolysis. Water offers a hydroxyl group (-OH) and hydrogen (-H), to both the monosaccharides caused breaking of glycosidic bond.
Examples of Disaccharides –
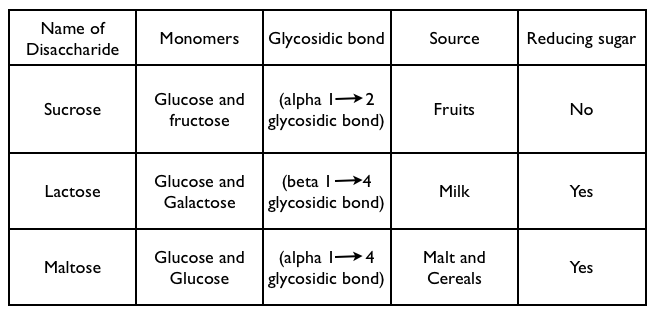
3.Polysaccharides
- Polysaccharides are polymers with combination of many monosaccharide molecules (more than two) by condensation reactions.
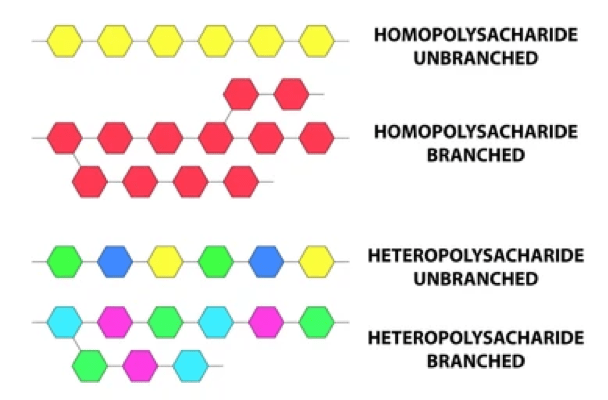
- Molecules with 3-10 sugar units are called “oligosaccharides” while molecules that contains 11 or more monosaccharides are true polysaccharides.
- Polysaccharides do not have sweet taste.
- Because of their large molecular size, the majority of polysaccharides do not dissolve in water.
- Polysaccharides made from one kind of monosaccharides are called “homopolysaccharides” (Starch) while those made of more than one monomer are called “heteropolysaccharides” (Hyaluronic acid).
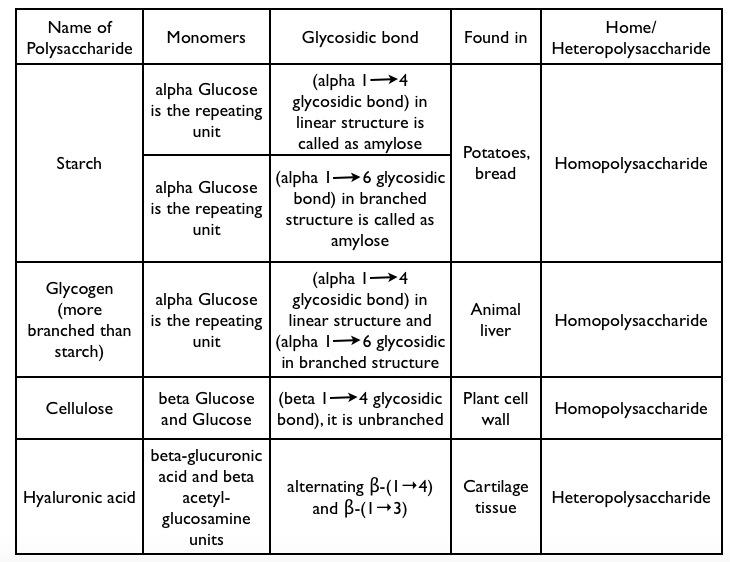
References –
https://dlc.dcccd.edu/biology1
https://med.libretexts.org/Courses/American_Public_University