Cultivation of the Anaerobic bacteria is challenging. It needs special media and incubation environment.
Introduction to Cultivation of the Anaerobic bacteria-
Based on the requirement of oxygen, the microbes are broadly classified as Aerobic and Anerobic. Aerobic organisms needs oxygen whereas, anaerobic microorganisms do not need oxygen. All eukaryotic organisms require oxygen to thrive, many species of bacteria can grow under anaerobic conditions. The aerobic life forms needs oxygen because it acts as a final electron acceptor in the Electron Transport Chain. When oxygen accepts the electron, it become highly excited and reactive. The reactive nature of the oxygen is controlled and suppressed by catalase. Such protective mechanism is absent in anaerobic microbes and hence they cannot tolerate oxygen.
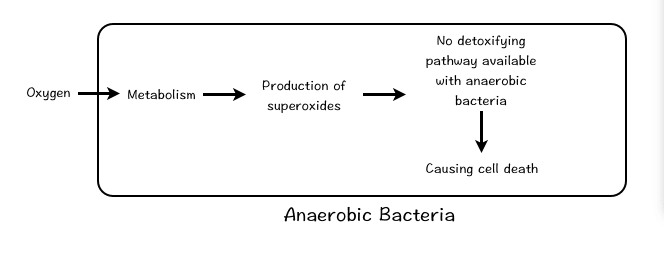
The aerobic and anaerobic microbes are further classified based on extent of oxygen requirement. Bacteria that can grow only in oxygen are called as obligate aerobic bacteria. Anaerobes, do not need oxygen for their growth and survival. Anaerobes are further categorize into three categories: Obligate anaerobes could be damaged or even destroyed by oxygen; Aerotolerant anaerobes do not use oxygen to live, however they are able to exist in its presence; and lastly Facultative anaerobes are aerobic microbes but in absence of oxygen, they can swtich to anerobic metabollic pathways (i.e. fermentation).
As oxygen is ubiquitous in air, retaining O2-free conditions during all steps of cultivation is challenging but a prerequisite for anaerobic culturing.
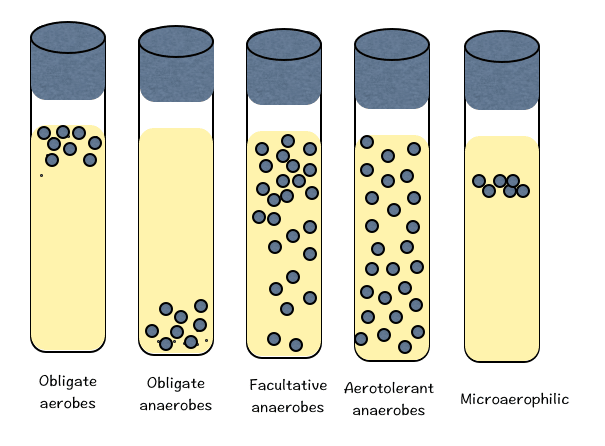
- Obligate aerobes grow only at the top of such tubes.
- Facultative organisms grow throughout the tube but best near the top.
- Microaerophiles grow near the top but not right at the top.
- Anaerobes grow only near the bottom of the tube, where oxygen cannot penetrate.
Principle of Cultivation of the Anaerobic bacteria–
Oxygen is ubiquitous in the air so special methods are needed to culture anaerobic microorganisms. Most strict anaerobes require not only the absence of oxygen to initiate growth, but also a redox potential below -300mV, which can be only achieved by the supplementation of media with reducing agents. Thioglycollate broth is a multi-purpose, enriched differential medium used primarily to determine the oxygen requirements of microorganisms. Sodium thioglycollate in the medium consumes oxygen and permits the growth of obligate anaerobes. This, combined with the diffusion of oxygen from the top of the broth produces a range of oxygen concentrations in the media along its depth. The oxygen concentration at a given level is indicated by a redox sensitive dye like resazurin that turns pink in the presence of oxygen. The main principle is to reduce the O2 content of culture medium and remove any oxygen already present inside the system or in the medium.
Requirements:
- Bottles or tubes filled with culture with stopper
- Reducing agent
- Thioglycollate broth
- Redox indicator dye
Composition of Thioglycollate media (pH) – 7.1 ± 0.2
| Ingredients | Gms/Liter |
| L – cystine | 0.50 |
| Sodium chloride | 2.50 |
| Glucose | 5.50 |
| Yeast extract | 5.00 |
| Pancreatic digest of casein | 15.0 |
| Sodium Thioglycollate | 0.5 |
Procedure –
- Add the ingredients of Thioglycollate medium in one liter of Distilled water.
- Boil the media in order to dissolve the ingredients of the media.
- Suspend the media in the cap tubes and autoclave at 121°C for 15 minutes.
- Collect some soil sample from eluviation layer. Dissolve it in water.
- Dilute the dissolved soil sample serially.
- Inoculate the serially diluted sample in the sterilized capped tubes. Closed the caps tightly.
- The inoculated media is incubated in anaerobic chamber or jar.
Result –
Reducing media chemically remove molecular oxygen (O2) that might interfere with the growth of anaerobes. Thioglycollate combines with dissolved O2 to deplete its concentration in the media allowing anaerobic bacterial growth.
Conclusion –
It is difficult to grow anaerobic bacteria as compared to aerobic bacteria. Thioglycollate media seems to work efficiently in providing anaerobic environment.
References –


One thought on “Cultivation of Anaerobic Bacteria – Thioglycollate medium”