Introduction to Acid Fast Staining-
- In 1882, Paul Ehrlich developed the Acid Fast Staining technique.
- Later, Ziehl and Neelson developed it in 1883 and hence the technique is also called as “Ziehl-Neelsen Staining Technique”.
- The main aim of this technique was to distinguish bacteria between acid fast groups and non-acid fast groups.
- Acid-fast organisms are characterized by wax-like nearly impermeable cell wall, containing mycolic acid along with large amount of fatty acids, waxes, and complex lipids.
- The acid-fast microorganisms are resistant to decolorization by acid due to the composition of cell wall.
- This test is exclusively used for identification of Mycobacterium tuberculosis, a causative agent of TB. It is a member of Mycobacterium genus.
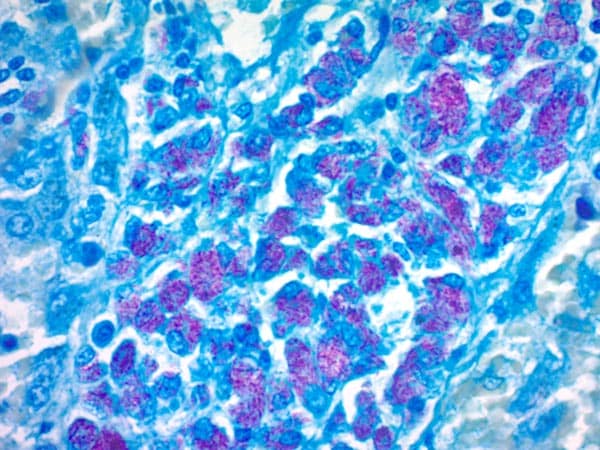
- In Ziehl & Neelson staining method, the acid-fast bacteria appear to be red bright bacilli with a light blue background.
Need of Acid Fast Staining-
- The Gram staining method cannot distinguish all types of bacteria.
- There is group of bacteria, which gets poorly stained with gram staining.
- Hence, we need an alternative method.
- Acid-fast staining is a special type of staining method used for the organisms that are resistant to acid.
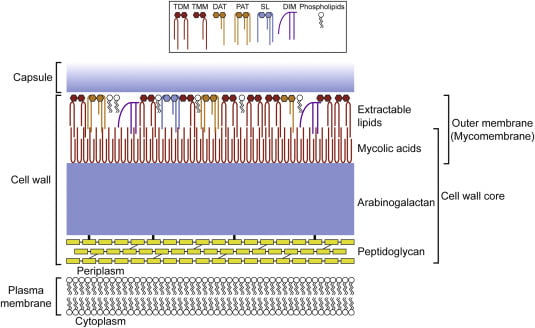
Composition of cell wall of Acid Fast Bacteria-
Before understanding the principle, it is important to understand the composition of acid fast bacterial cell wall. The acid-fast bacterial envelope consists of 5 layers. The outer layer is made up of mycolic acid, waxes, lipid and polypeptides (layer 1). The presence of mycolic acid makes it resistant from many chemicals and phagocytosis. The surface protein or polypetides are mostly involved in adhesion. It is followed by thick peptidoglycan (layer 2) which is similar to gram-positive bacteria. Its function is to provide protection from osmotic pressure. The peptidoglycan is linked to arabinogalactan (layer 3), it provides strength to the cell wall. Layer 4 is the periplasmic space present between the cell wall and cytoplasmic membrane. It is rich with metabolic enzymes. Layer 5 is the cytoplasmic membrane.
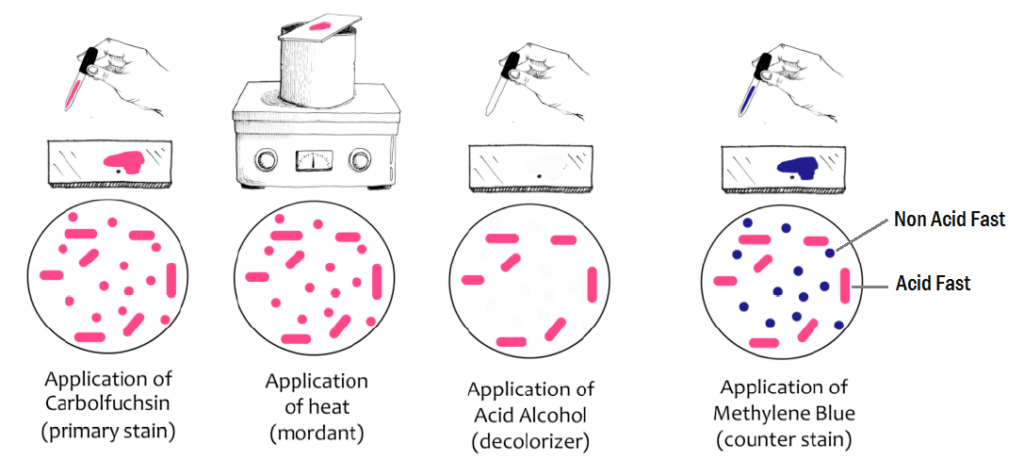
Principle –
When the bacterial smear is flooded with carbol fuschin (primary stain) in presence of heat, the stain penetrates in the bacterial cell wall. Due to which bacteria appears red in color. Due to the presence of mycolic acid and high lipid content, even after decolorization step, the acid fast bacteria retains the primary stain whereas the non acid fast bacteria gets decolorize. When the smear is flooded with methylene blue (counter stain), the decolorized non acid-fast bacteria takes the counter stain and appears blue in color. Whereas, the acid fast stain bacterial group cannot taken the counter stain because of the presence carbol fuschin stain in their cell wall and hence they appear red in color.
Reagents required –
- Ziehl Neelson Carbol – fuchsin (primary stain).
- Acid Fast Decolourisation (acid alcohol)
- Methylene Blue (secondary stain – 0.3%)
Apparatus required –
- Acid fast kit (includes above mentioned reagents)
- Bunsen Burner
- Slides
- Glass rods
- Inoculating loop
- Mycobacteria culture
- Microscope (oil immersion objective)
Procedure –
- Prepare a smear on clean grease free slide.
- Allow it for air dry and fix it by giving a gentle heat.
- Flood the slide with Carbol Fuchsin stain until it get spread all over the slide.
- Heat the slide for 5 minutes on a very low flame (temperature may go up to 60°C.
- Then the slide is kept undisturbed for 5 minutes and allows it to cool.
- Rinse the slide with distilled water and wash the stain.
- Washing is followed by Acid Fast Decolourizer (3% v/v acid alcohol) treatment for about 2 minutes.
- Remove the Acid-fast Decolourizer with water.
- Flood the slide with Methylene Blue (counterstain) for about 2 min.
- Wash the counter stain, allow it dry in air and then observe it under oil immersion objective.
Note-
- Do not boil the stain.
- While performing the staining, the smear should not be dry.
- Use distilled water for washing.
Examples –
ACID-FAST BACTERIA – Mycobacterium tuberculosis, M.phlei, M. Leapre, etc.
NON-ACID FAST BACTERIA – E.coli, Staphylococcus aureus, etc.
Advantages –
- It is very useful in quick detection of infectious cases of pulmonary TB.
- It is used to distinguish Mycobacterium genus with other bacteria.
- It is simple to perform
- It is a cheap method
Disadvantages –
- The method cannot distinguish between Mycobacterium tuberculosis from Non Tuberculosis Mycobacterium.
- It cannot distinguish living and dead cells.
- If in-vitro acid fast cultured cells are stained, they may get poorly stained because of low lipid content.
Modifications –
The conventional method is still being modified in order to overcome the limitation and disadvantages of Ziehl-Neelsen method
- KINYOUN MODIFICATION – Also known as Cold Ziehl-Neelsen Technique is also available.
- A new protocol is created in which the “toxic phenol” in fuchsin has been replaced by a detergent.
- Variable Differential Staining – In this, glacial acetic acid is used with no heat and where the secondary stain is Loeffler’s Methylene Blue.
Applications –
- It is used to examine and identify the members of genus Mycobacteria.
- It is used for identification of some fungal species such as Cryptosporidium.
Conclusion –
The acid-fast bacteria appears in red color because they retain the primary stain whereas the non acid fast bacteria appears blue color as they take methylene blue.
References –
https://en.m.wikipedia.org/wiki/Ziehl%E2%80%93Neelsen_stain

It’s good and knowledgeable for who study in biology field.