Stem cells are the progenitor cells that further differentiate into different types of cells. They are called the foundation for forming tissue, organs and body. The stem cells maintain their population and renew themselves by cell division. There are different types of stem cells based on the ability to form a range of differentiated cells.
Totipotent – ability to form all cell types required for body formation and placental cells.
Pluripotent – ability to form all different types of cells required to form a whole organism (not placental cells).
Multipotent – Ability to form more than one type of cells.
Unipotent – ability to form only one type of cell.
In blood, all the cells are formed from stem cells called Hematopoietic stem cells (HSC) hence they are examples of pluripotent cells. The process of forming blood cells from HSC is called as Hematopoiesis. In human growth developmental stage, the blood cells are formed in different ways. In the first week of embryo developmental stage, the blood cells are formed from yolk sac stem cells. Such cells contain embryonic haemoglobin. In a three month foetus, the hematopoietic stem cells travel to the liver and spleen and further these two organs play a major role in blood cell formation. In the 7th month, the hematopoietic stem cells settle in bone marrow and become sites for Hematopoiesis. After birth, the spleen and liver are no longer involved in blood cell formation.
| Developmental stage / age | Site for Hematopoiesis |
| Embryo | Yolk sac |
| 3 months foetus | Spleen and liver |
| 7 months foetus | Bone marrow; spleen and liver if needed |
| After birth | Bone marrow only (skull, ribs, sternum, vertebral columns, pelvis and femurs) |
Renewing ability of Hematopoietic cells –
With the advance technology in in vitro lab conditions, we are able to understand and learn about stem cells growth and its differentiation process. But still our knowledge is limited and we still need to learn more about its regulation. Ability to renew HSC was demonstrated in mice. A normal mouse contains 3 x 108 cells, but with the X rays lethal dose, 90% of hematopoietic stem cells were killed, such mice cannot live more than 10 days. Such mice were saved by transplanting 104 to 105 bone marrow cells from genetically identical mice. Translating just 0.01 to 0.1% of hematopoietic cells were found to be sufficient to restore and replenish the loss. This experiment witnessed the renewable ability of HSC.
Scientists consider HSCs as one of the standard models to study stem cells and tissue systems and to study their roles in causing ageing and cancer.
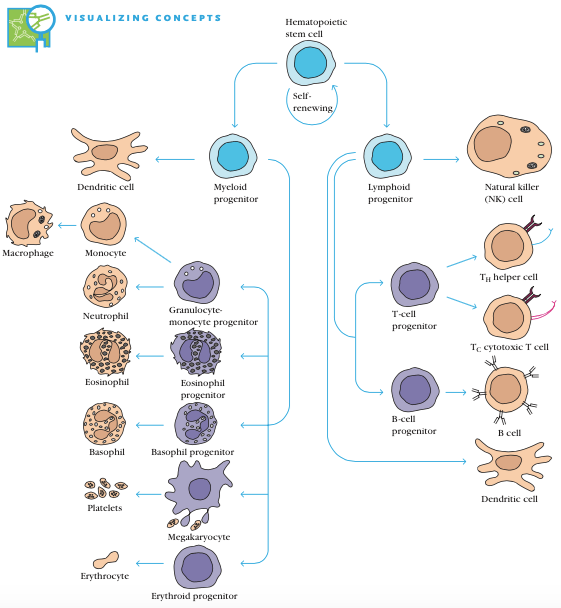
A multipotent Hematopoietic cell has the ability to form different blood cells. The process is initiated by forming two major progenitor cells – Lymphoid or Myeloid. The Lymphoid progenitor cells differentiate into Natural Killer cells, T cells (Tc and TH), B cells and Dendritic cells whereas the Myeloid progenitor cells differentiate into Dendritic cells, Granulocyte monocyte progenitor, Eosinophil progenitor, Basophil progenitor, Megakaryocyte and Erythroid progenitor. These progenitor cells are further differentiated into Monocyte & Macrophages, Neutrophil, Eosinophil, Basophil, platelets and Erythrocyte respectively.
Regulation of Hematopoiesis by cell communication –
The process of self renewal and differentiation of HSC is regulated and controlled by various growth factors. The growth factors like cytokine belong to colony stimulating factors (CSF) and glycoprotein erythropoietin (EPO). Research showed that EPO is involved in erythrocyte’s growth and development. Further it has been shown that there is a specific relationship between signal and receptor of target cell. The binding of signal to target receptor causes signal transduction that leads to regulation of molecular pathways like gene expression and protein synthesis.
The site for Hematopoiesis is bone marrow, where hematopoietic stem cells grow and mature on non hematopoietic cells called stromal cells. Macrophages, fat cells, fibroblasts, endothelial are some examples of stromal cells. The stromal cells provide an ambient environment to HSC for its growth and differentiation by releasing hematopoietic-inducing microenvironment (HIM). The cell communication between stromal cells and hematopoietic cells can be direct (cell to cell contact) or indirect in the form soluble agents that can diffuse into the target cell. The cell communication between stromal cells and HSC is affected by the health status of an individual. Research has found that in an infected person, their cell communication is stimulated by the growth factors produced by the macrophages and T cells.
Due to the relationship of stromal cells and HSC, we were able to grow them in in vitro conditions. In vitro studies allowed us to study and find the hematopoietic growth factors involved in hematopoiesis. In vitro, the lymphoid and myeloid cells were cultured with nursing stromal cells providing a microenvironment for HSC growth and proliferation. The growth is visible in the form of colonies.
Regulation of Hematopoiesis by genes-
Using knockout technology, the studies have found that a set of particular genes are involved in regulating the hematopoiesis process. Knockout technology is a powerful technique in which in an organism a gene or few genes are made inoperative. This technique is useful for studying the role of particular genes in molecular pathways. Using this technique, it was observed that GATA-2 gene plays an important role in development of lymphoid, erythroid and myeloid lineages. The GATA-2 gene produces a transcription factor that regulates the development of lymphoid, erythroid and myeloid cells. Knockout technology also showed that Ikaros is another transcription factor involved in development of lymphoid cell lineage like T cells, B cells and Natural killer cells. However, the impaired Ikaros gene does not affect the development of myeloid cell lineage.
Population of blood cells is regulated –
The hematopoiesis maintains the steady population of blood cells. The life span of blood cells are different, for example the erythrocytes lifespan is 120 days whereas T lymphocytes live for 20 to 30 years.
| Blood Cells | Life span |
| Neutrophil | 7 hours |
| Eosinophils | 8 to 12 days |
| Basophil | May be for few hours for for fee days |
| Monocyte | 3 days |
| B cells and T cells | Weeks to years |
The steady state population is maintained by controlling the production and release of cytokines and by regulating the expression of receptor proteins on the target cells. The cell population is also controlled by programmed cell death called apoptosis.
References-
Kindt, T., Goldsby, R., Osborne, B., Kuby, J. and Kuby, J., 2007. Kuby immunology. New York: W.H. Freeman.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3666375/
Dr. Sangha Bijekar has 9 years of Teaching Experience at University level. She loves to get engage in teaching and learning process. She is into blogging from last two years. She intends to provide student friendly reading material. She is avid Dog Lover and animal rescuer. She is learned Bharatnatyam and Katthak Dancer. She is into biking and She also loves to cook.