A quick read up on DNA to refresh those brain cells. Jog your memory cells down the DNA strand.
Different Forms of DNA and the Structure of DNA are by far the most asked questions by the students who are browsing for a quick read as tomorrow is the Practical/Viva. A handy, easy to read answer is what students look out for. The following article aims to do exactly that! Help out a student to know the different forms of DNA.
DNA is a polymer of nucleotides. The repeating unit of polynucleotide chain is Nucleotides; they are composed of sugar, phosphate, and nitrogen bases. The nucleotide structure is held by glycoside bond and ester bond. The glycoside bond is present between sugar (C1) and nitrogen base (N1 in Pyrimidine and N9 in Purine) and ester bond is present sugar (C5’) and phosphate. The two polynucleotide strands are held together by hydrogen bonds formed between the complementary Nitrogen base pairs. The other non-covalent bonds like hydrophilic, hydrophobic, Vander wall, and ionic bonds also contribute in maintaining the native conformation of DNA.
The DNA is found to be in different forms because of its flexibility. The sugar phosphate backbone allows considerable rotation at bond. Thermal variation also causes stretching or bending or unpairing of the strands. The flexibility of bond rotation and thermal fluctuation allows DNA to have different forms. The physiochemical environment dictates the formation of these different forms.
The glycoside bond present between C1′ of sugar and N1 or N9 of nitrogen base is found to be flexible. The sugar and nitrogen base’s plane are almost perpendicular to each other. And that allows the formation of two different conformation of nucleotides called as “Syn” and “Anti”. In Anti-conformation, H6 of purine or H8 of pyrimidine atoms are above the sugar whereas in “Syn” conformation, N3 of Purine and O2 of pyrimidine. In A and B form of DNA, the purines and pyrimidines are found in anti-conformation. The purines are found to be in syn conformation in Z form of DNA.

X ray crystallography studies have found that there are three different forms of DNA which are the A, B and Z forms.
The B form is referred to Watson and Crick model. It is considered as benchmarks or standard for conducting DNA studies.
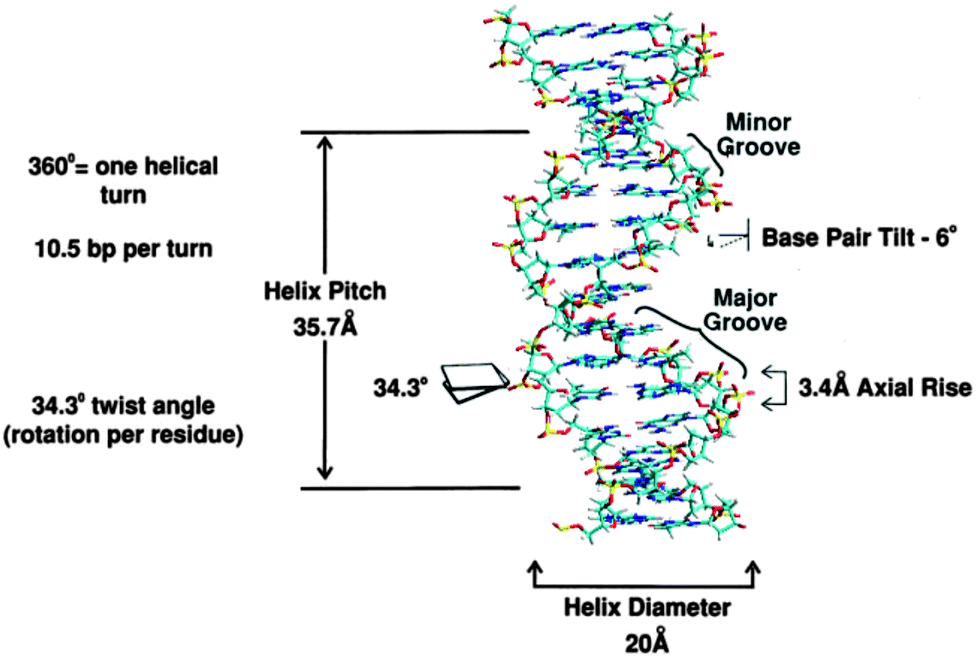
The characteristics of B form of DNA are-
- It consists of two strands and both the strands are intertwined in right-handed direction forming right-handed helix.
- The both the strands are antiparallel to each other, one runs from 5’ to 3’ and other runs from 3’ to 5’.
- The both strands are hold together with Hydrogen bonds because of complementary relationship.
- The purine and pyrimidine are in anti-conformation at glycosyl bond.
- The diameter of B form of DNA is 20 Å.
- The length of helix pitch is 34 Å.
- One helical turn contains 10 bp.
- The helix rise is 3.4 Å, it the distance along the helical axis between nucleotides.
- The puckered sugar conformation is C2’ endo type.
- The base pair tilt is 6°.
- The major grooves are wide and deep; minor groves are narrow and shallow.
The characteristics of A form of DNA are-:
- It is right handed helix, and both the strands are held together by hydrogen bonds.
- Both the strands are antiparallel in nature.
- The purine and pyrimidine are in anti-conformation at glycosyl bond
- The diameter of A form of DNA is 26 Å.
- The length of helix pitch is 28 Å.
- One helical turn contains 11 bp.
- The helix rise is 2.6 Å is helix rise.
- The puckered sugar conformation is C3 endo type.
- The base pair tilt is 20°.
- The major groves are deep and narrow; minor groves are shallow and wide.
Reason for the formation of A form of DNA:
The A form of DNA is formed in a dehydrated environment or in less than 75% humidity. The sugar phosphate backbone is hydrophilic, and the interior part of helix i.e. nitrogen bases are hydrophobic. The hydrophobic nitrogen base pairs group together and tend to stay away from the aqueous environment. The dehydrated environment allows them to open up thus widening the diameter of DNA.
The existence of A form in live cells is not certain. A form of DNA is formed during crystallization step of X ray studies, as crystallization promotes dehydration forming A form.
The characteristics of Z form of DNA are:
- It is left handed helix and both the strands are hold together by hydrogen bonds.
- The phosphodiester bonds are on zig zag pattern and hence named as ‘Z’
- The alternate purine and pyrimidine sequence in DNA favors the formation of Z form Ex. GCGCGCGCGC.
- Both the strands are antiparallel in nature.
- The purines (G) are syn and pyrimidines (C) are in anti-conformation at glycosyl bond (reason for left handed).
- The puckered sugar conformation is C2’ endo for pyrimidines and C3’ endo for purines (reason for left handed).
- The diameter of Z form of DNA is 18 Å.
- The length of helix pitch is 45 Å.
- One helical turn contains 12 bp.
- The helix rise is 3.7 Å.
- The base pair tilt is 7°.
- The major groves are almost flat; minor groves are narrow and deep.
- It is formed at small stretch of whole DNA; it is formed during gene expression or recombination of genes.
Bonus Activity for understanding the different forms of DNA
Based on the information shared above, distinguish the different forms of DNA
| A form | B form | Z form | |
| Helix direction | |||
| Diameter | |||
| Number of base pair per helical turn | |||
| Base tilt normal to the central axis | |||
| Sugar puckering conformation | |||
| Glycosyl bond conformation | |||
| Grooves |
Meme
References:
- Biochemistry, 5th editionJeremy M Berg, John L Tymoczko, and Lubert Stryer.New York: W H Freeman; 2002.
- David Hames and Nigel Hooper (2005). Biochemistry. Third ed. Taylor & Francis Group: New York.
- Nelson, David L. Lehninger Principles Of Biochemistry. New York : W.H. Freeman, 2008.
- https://www.nature.com/scitable/definition/phosphate-backbone-273
- Bailey, W. R., Scott, E. G., Finegold, S. M., & Baron, E. J. (1986). Bailey and Scott’s Diagnostic microbiology. St. Louis: Mosby.
If you liked this resource, please Like, Share, and Subscribe us for more content.
Dr. Sangha Bijekar has 9 years of Teaching Experience at University level. She loves to get engage in teaching and learning process. She is into blogging from last two years. She intends to provide student friendly reading material. She is avid Dog Lover and animal rescuer. She is learned Bharatnatyam and Katthak Dancer. She is into biking and She also loves to cook.



One thought on “Different Forms of DNA | A Quick Read | BiokiMicroki”