Introduction
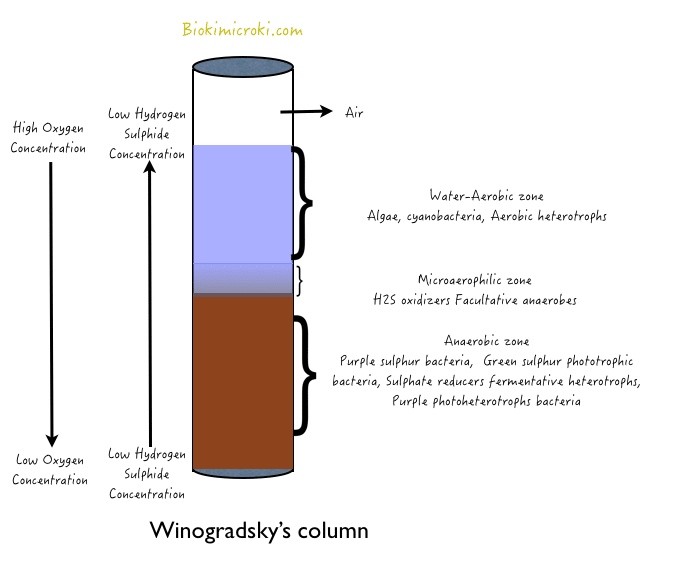
Winogradsky’s column is used to study cross section of a natural environment in the laboratory. Sergei Winogradsky invented the column in the 1880s. Various ecosystems such as sewage, pond water, soil can be represented in column. It is also used to isolate various photosynthetic bacteria such as purple and green photosynthetic bacteria along with sulfur reducers and many other anaerobic bacteria. This column is prepared by filling a glass cylinder or a transparent clean bottle with soil and mud sample till half of the cylinder supplementing with shredded filter paper, news papers (source of carbon), CaCo3, blackened marshmallows or egg-shells containing calcium carbonate , K2HPO4(buffer), and CaSo4 or egg yolk (source of sulfur). To this water sample of the area whose ecosystem is to be studied is filled till top of the column and cover the opening tightly to prevent evaporation. After incubation of about 3-6 weeks in area having sufficient sunlight, various successions of organisms develop depending on the availability of light, oxygen and concentration of nutrients.
While the Winogradsky column is an excellent tool to see whole communities of bacteria, it does not allow one to see the densities or individual bacterial colonies. It also takes a long time to complete its cycle. However its importance in environmental microbiology cannot be overlooked and it is still an excellent tool to determine the major bacterial communities in a sample.
Principle –
Winogradsky’s column has organic materials which triggers the microbial activity resulting in the oxygen depletion from soil and hence creating a gradient of low oxygen at bottom and high oxygen at top of the column. Due to these gradient, anaerobic conditions lead to the growth of anaerobic bacteria including Clostridium sps. Filter paper added in the column is made up of cellulose which breaks down to glucose subunits hence providing source of carbon and energy to the microbial population and resulting to the production of ethanol and organic acids as by-products through their metabolism process. Organic acid produced as a by-product is used by other bacteria like Desulfovibrio as a source of carbon whose growth inturn leads to H2S production, as sulfur acts as terminal electron acceptor in respiration of the organism. H2S produced in anaerobic condition hence its concentration will be high at the bottom of the column creating H2S gradient.
In Winogradsky column, H2S is metabolized by photosynthetic bacteria such as Chlorobium (green sulfur bacteria) and Chromatium (purple sulfur bacteria). They use CO2 as carbon source, H2S as electron donor and light as energy source and produces organic compounds. As Purple bacteria are low tolerant to H2S concentration, they grow away from the source of H2S production. They are normally found at mid region of the column. Other than these purple non-sulfur bacteria such as Rhodomicrobium grows using ethanol as electron donor.
At interface of soil and mud, aerobic sulfur oxidizers such as Beggiatoa grows using H2S as energy source and oxidizes it to H2SO4. They fix CO2 to organic compound. In water layer, aerobic photosynthetic organisms are abundant such as Cyanobacteria and Algae, which produces oxygen as by-product helping in maintaining oxygen gradient. Thus each layer of Winogradsky’s column supports certain types of bacteria, which dominate the culture at that region hence helping in studies of a particular ecosystem.
Requirements:-
- Soil sample.
- Shredded filter paper.
- CaCo3, K2HPO4, CaSO4.
- Water sample.
- Pipettes.
- Saline tube/ glass cylinder or jar/ transparent bottle/PET bottle.
- Plastic cover paper.
Procedure:-
- Fill the clean transparent bottle or glass cylinder with shredded filter paper to about 1-2 inches.
- Mix 1gm of CaS04 and 500mg K2HPO4 with small amount of soil sample and fill in the cylinder up to 3-4 inches level over the shredded filter paper.
- Fill the cylinder with water sample to about 0.5 inch from the top.
- Cover the top of the cylinder with plastic cover paper to prevent evaporation.
- Incubate the column for 2-4 weeks exposed to sunlight.
- Observe the changes that occur, during this period as microbial population develops in zone that suits a particular environment.
- Record the observations as photographs for comparison results.
- Study the micro flora of different zones by gram staining and wet mounting.
- Remove the water sample from various levels using pipette with a large tip orifice, holding your index finger firmly over the end and carefully inserting pipette into the level, which is to be examined. Remove your finger and allow a small amount of water at that level to enter the pipette then close the tip again with the finger to lock the sample in pipette. Carefully remove the pipette without disturbing other zones or layers of the column.
- Remove soil sample by de mantling the column by slicing the bottle at different levels. This is done when column is made in PET bottle.
- Samples from the column can be used for enriching various groups of microorganisms.
Results:-
1. Winogradsk’s column was prepared and results as shown in the picture (i) is obtained after 4 weeks of incubation in sunlight. Which shows blackening at the bottom due to H2S production, pinkish purple layers above the sediments, green growth at walls of bottle which is exposed to sunlight and surface turbid layer.
2. Wet mount was plotted from the deep layer of water from column resulted showed oval structured organisms arranged in chains.
3. Wet mount was plotted from the surface turbid layer of water from walls of cylinder or bottle with water from column showed various filamentous structures.
Interpretation and conclusion:-
Winogradsky’s column was prepared to study various successions of variety of organisms involved in metabolism of sulfur. The provable organisms observed belong to sulfur reducing bacteria such as Desulfovibrio and sulfur oxidising bacteria such as purple sulfur bacteria, Cyanobacteria, green algae, protozoa etc. Further confirmation of the organisms can be done by gram staining , culturing and identifying the organisms from different layers of the column.
References:-
Corner, T. (1992). Ecology in a Jar. The Science Teacher. (59)3, 32
Zavarzin, G.A. (2006). Winogradsky and Modern Microbiology. Microbiology. (75) 5, 501-511
biofilmbook.hypertextbookshop.com


Thank you for sharing the knowledge. It will be very helpful for students like us.
Easy and concise informatio.