Paper Chromatography is an analytical technique used for separation, detection and identification of biomolecules.
Chromatography is a fundamental technique for the separation, detection, identification of chemical species from a mixture. As it is called as analytical technique because it is used for qualitative and quantitative studies of separated components from mixture. The chromatography consists of two phases – Stationary and Mobile phase. The name of the phases suggest its function. The stationary phase remains still while the mobile phase move. The mobile phase moves through stationary phase.
When we isolate organic or biomolecules from natural sources, they are mostly present in the form of mixture. In order to study the biological molecules we need them to separate them from the mixture.
To learn the fundamentals of chromatography read our article on principle, types and application of chromatography.
Paper Chromatography–
It is planar chromatography. It is one of the simplest types of chromatography. The principle of paper chromatography is partition. In paper chromatography, the paper acts a stationary phase and organic solvent or mixture of solvents is mobile phase.
Principle of paper chromatography–
When the mixture (sample) is loaded on chromatography, the different components of mixture interacts differently with stationary and mobile phase. The affinity of components of mixture for stationary and mobile phase is different. When mixture or sample is loaded on chromatography, the components of mixture travels up on stationary phase with the mobile phase. The developing solution travels up the stationary phase carrying the sample with it. Components of the sample will separate readily according to how strongly they adsorb onto the stationary phase versus how readily they dissolve in the mobile phase.
If components have high affinity for mobile phase then they tend to spend more time with mobile phase than stationary and hence they travel more than the components which have high affinity towards stationary phase. Due to the difference in affinity for stationary and mobile phase, the components travels with different speed and gets separated.
Characteristics of paper-
The paper in the paper chromatography is cellulose. Cellulose is a polymer of glucose. The OH groups of the glucose protrude out from the cellulose polymer. The cellulose paper holds moisture in its pores or space present between the networks of cellulose fibers of paper. The presence of moisture in paper makes it polar and allows it form non covalent bonds with the polar components or compounds. For paper chromatography, different grades of Whatman filter papers are used.
Procedure–
- Selection of solvent system – Select appropriate solvent or mixture of solvent as mobile phase. The mobile phase is selected based on the sample and aim of separation of components from mixture.
- Selection of Stationary Phase – Select appropriate paper for chromatography. The paper would act as a stationary phase.
- Preparation of Sample – sample may be required to dissolve in suitable solvent. (ex. Leaves are crushed in mortal pestle using appropriate solvent)
- Loading Sample – The sample or mixture (whose components need to be separated) is loaded or spotted on chromatographic paper. It is loaded 2 cm above the bottom line.
- Chromatographic Chamber – The chromatographic chamber is made from stainless steel, plastic or glass. The glass chromatographic chamber is preferred more over other material because the transparent nature of glass allows us to keep an eye on developing chromatogram. Before placing the chromatographic paper, the chamber is saturated with the solvent or mobile phase. The paper is suspended in chamber containing appropriate solvent or mixture of solvent as a mobile phase. The distance travelled by mobile phase or solvent is called as solvent front.
- Chromatogram Development – Due to capillary action, the solvent rises and moves up against the gravitational force. While moving up, the mobile phase (solvent or mixture of solvent) flows over the loaded sample (Which appears like a spot). The components of the mixture (sample) will interact differently with stationary and mobile phase due to which the rate of movement of individual components will be different. The difference in the rate of movement would separate the all the components of mixture.
Hence, the principle of paper chromatography is partition because the components of mixture get partitioned due to different affinity towards stationary and mobile phase. The chromatographic paper on which different components have traveled and got separated is called as chromatogram. If the components of mixture are colored compounds, they look like colored bands. If the components are colorless, then appropriate spray or dye is used or may be observed under UV.
Identification of Sample –
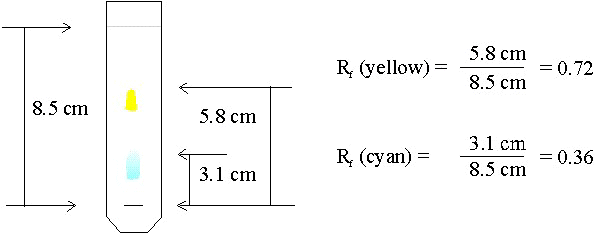
Due to difference in the affinity for stationary phase and mobile phase; different components travels to different extent. Some components travel as far as solvent move and few components travel less and remain close to spotting line. The distance traveled by the components in particular solvent system (mobile phase) always remains the same and hence can be used as a characteristic for identification of components of mixture or sample. The term that indicates how far the component or molecular has traveled from spotting line is called as Retention factor (Rf).
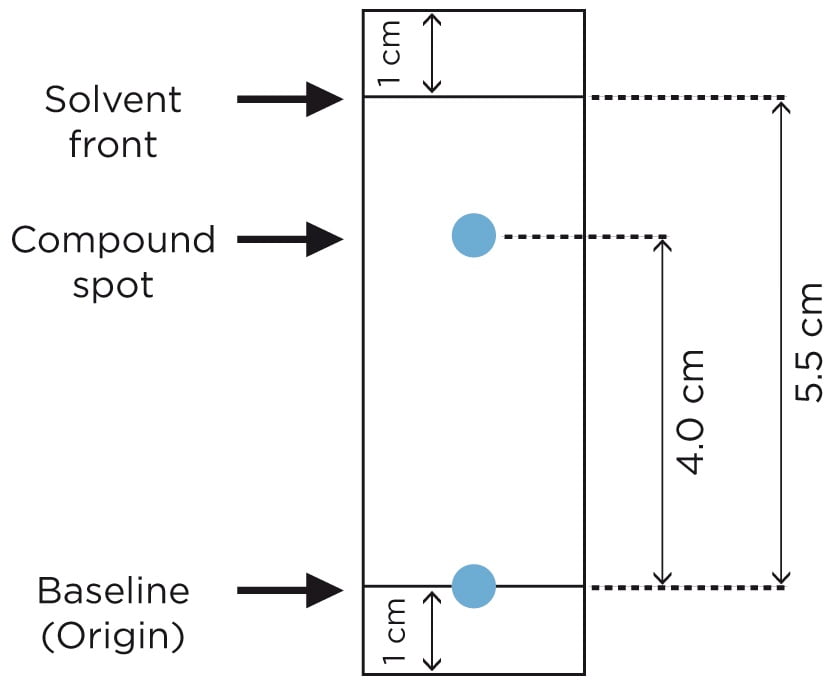
Retention factor or Rf value is the ratio of distance traveled by solvent front by distance traveled by the components or molecule. Hence to calculate the Rf, we need to measure the distance traveled by solvent front and components of mixture or sample.
The Rf value of biomolecule or compound is affected by following factors-
- Type or composition of mobile phase
- The quality or grade of paper
- Temperature
Application of Paper Chromatography-
- To identify the components of biomolecules
- To separate the biomolecule from the mixture
- It is used in separating pigments
- It is used for qualitative analysis
- It is employed for analyzing complex mixtures.
Advantages:
- It is quick and simple to perform
- It is cheap method
- It requires sample in low volume
- Due to its small size, it does not require much space.
- It can separate organic and inorganic molecules
- It can be used in different industries like cosmetics, drugs, food and agriculture etc.
Limitations:
- The efficiency of paper chromatography is poor as compared to other chromatographic techniques.
- It cannot conduct quantitative type of study
- It is comparatively less accurate.
References-
https://chem.libretexts.org/Bookshelves/Organic_Chemistry
https://pubs.acs.org/doi/abs/10.1021/ac60051a002
https://www.chemguide.co.uk/analysis/chromatography/paper.html
https://www.pulpand-technology.com/articles/
https://www.chemguide.co.uk/analysis/chromatography/paper.html
https://www.khanacademy.org/science
http://www.marz-kreations.com/Chemistry/Chromatography/Dyes/RF-Values.html



One thought on “Principle & Procedure of Paper Chromatography”